AstraZeneca
AZN
announced that the European Medical Agency (EMA) has authorized Vaxzevria, its COVID-19 vaccine, for use as a third/booster dose in adults in Europe. A primary two-dose vaccine regimen of Vaxzevria is already authorized for use in the EU.
Vaxzevria is now approved for use as a booster dose in adults who already completed a primary vaccination regimen with Vaxzevria or an mRNA-based COVID-19 vaccine developed by
Pfizer
PFE
/
BioNTech
BNTX
and
Moderna
MRNA
, both of which are already approved in the European Union (EU).
The EMA’s authorization follows a positive review by the Committee for Medicinal Products for Human Use (CHMP), which recommended the use of a booster dose of the vaccine. The decisions by both the EMA and CHMP are based on clinical studies, which demonstrated an increase in immune response in participants who were given the booster dose. These participants had already completed a primary two-dose vaccine regimen with Vaxzevria or mRNA-based vaccines approved for use in the European Union (EU).
In the year so far, shares of AstraZeneca have risen 14% compared with the
industry
’s 5.1% increase.
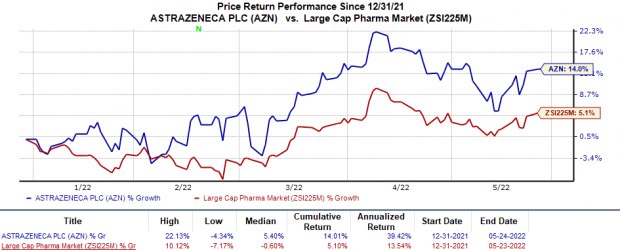
Image Source: Zacks Investment Research
Excluding the EU, AstraZeneca’s COVID-19 vaccine, either as a primary regimen or a booster dose or both, is already authorized for use in multiple countries across the globe. In fact, the primary vaccine series of the vaccine is granted authorization in more than 125 countries. However, AZN is yet to make a regulatory filing for Vaxzevria in the United States.
Last November, in the United States, the first booster doses of Pfizer/BioNTech and Moderna’s vaccines were granted an EUA for all adults (18 years of age and older). Later, Pfizer/BioNTech’s EUA was extended to allow the dosage in individuals aged 16-17 years and thereafter in adolescents varying from 12 years to 15 years of age. Last week, the Pfizer/BioNTech vaccine
was also granted
an EUA for use in kids between 5 years and 11 years of age.
In March this year, the FDA authorized a second booster dose of Pfizer/BioNTech and Moderna’s vaccines for older adults and some immunocompromised people. Pfizer/BioNTech and Moderna’s booster shots are authorized for use in adults in Europe.
Zacks Rank
AstraZeneca currently carries a Zacks Rank #3 (Hold). You can see
the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here
Zacks Names “Single Best Pick to Double”
From thousands of stocks, 5 Zacks experts each have chosen their favorite to skyrocket +100% or more in months to come. From those 5, Director of Research Sheraz Mian hand-picks one to have the most explosive upside of all.
It’s a little-known chemical company that’s up 65% over last year, yet still dirt cheap. With unrelenting demand, soaring 2022 earnings estimates, and $1.5 billion for repurchasing shares, retail investors could jump in at any time.
This company could rival or surpass other recent Zacks’ Stocks Set to Double like Boston Beer Company which shot up +143.0% in little more than 9 months and NVIDIA which boomed +175.9% in one year.
Free: See Our Top Stock and 4 Runners Up >>
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days.
Click to get this free report









