This week,
Pfizer
PFE
,
AstraZeneca
AZN
and
Glaxo
GSK
announced fourth-quarter and full-year 2021 results. While the FDA approved
Sanofi
’s
SNY
new rare blood disorder medicine, Enjaymo, an FDA advisory committee, voted that
Eli Lilly
LLY
and partner Innovent Biologics conduct an additional study to get approval for their lung cancer candidate, sintilimab, in the United States.
Recap of the Week’s Most Important Stories
Earnings Update:
Pfizer’s
fourth-quarter results were mixed
as it beat estimates for earnings but missed the same for sales. Direct sales and alliance revenues from it and partner BioNTech’s COVID-19 vaccine, Comirnaty, were $12.5 billion in the quarter,
Pfizer issued its financial guidance for 2022. Revenues are expected in the range of $98.0 billion to $102.0 billion. The revenue guidance includes approximately $32.0 billion in sales from Comirnaty and $22 billion from a new promising oral antiviral pill for COVID-19, Paxlovid. Adjusted earnings per share are expected to be in a range of $6.35 to $6.55. Its sales guidance for 2022 was below market expectations.
Glaxo
beat estimates for both
earnings and sales. While Pharmaceuticals sales were up 25% at CER, sales in the Consumer Healthcare segment increased 10%. Vaccine sales declined 7% at CER due to lower revenues from the majority of its vaccines, including Shingrix. Glaxo announced its earnings growth expectations for the year for the new company to be formed after the demerger of the Consumer Healthcare business. The company expects sales of the new Glaxo to increase 5% to 7% in 2022
AstraZeneca’s
fourth-quarter results were strong
as it beat estimates for both earnings and sales. Product sales growth was driven by its COVID-19 vaccine and higher sales of newer medicines across Oncology and CVRM units like Tagrisso and Farxiga, which offset some negative impacts from the pandemic and slower sales growth in China. The rare disease drugs added following the Alexion acquisition also aided sales growth.
AstraZeneca also issued encouraging guidance for 2022. It expects total revenues to increase in high-teens percentage in 2022.
FDA Approves Sanofi’s New Rare Blood Disorder Drug:
The FDA
granted approval
to Sanofi’s Enjaymo (sutimlimab-jome) for the treatment of hemolysis in adult patients with cold agglutinin disease (CAD), a rare blood disorder. Enjaymo is now the first and only FDA- approved therapy for treating patients with CAD. Regulatory applications seeking approval for sutimlimab in Europe and Japan are currently under review. In November last year, the FDA had
issued
a complete response letter (CRL) to the biologics license application (BLA) for sutimlimab due to certain deficiencies identified by the FDA during a pre-license inspection of a third-party manufacturing facility. Finally, the FDA accepted the resubmission of the BLA for sutimlimab in October 2021.
The FDA accepted and granted priority review to Sanofi’s supplemental biologics license application (sBLA) seeking approval for its blockbuster drug Dupixent for treating moderate-to-severe atopic dermatitis in children aged 6 months to 5 years. The FDA’s decision is expected on Jun 9, 2022. Presently, Dupixent is approved to treat moderate-to-severe atopic dermatitis in patients 6 years and older.
FDA Panel Votes Against Approving Lilly’s Sintilimab BLA:
The FDA’s Oncologic Drugs Advisory Committee voted 14-0 that Lilly and partner Innovent Biologics conduct an additional trial for seeking approval of their lung cancer candidate, sintilimab
Lilly/Innovent market sintilimab with the trade name of Tyvyt only in China. Lilly is seeking approval for sintilimab in the United States for the first-line treatment of adult patients with advanced non-squamous non-small cell lung cancer (NSCLC).
The ODAC committee’s negative vote stemmed from the fact that the BLA for sintilimab, which was submitted by Innovent, was based on a study, ORIENT-11, conducted exclusively in China. The committee argued that the data from the study are not applicable to the U.S. population and U.S. medical practice. It is difficult to make a definitive conclusion if the pharmacokinetics (PK) data can be applied to the diverse U.S population. The committee thus recommended an additional study to demonstrate applicability to the U.S. population before the BLA can be approved. Lilly and Innovent stated their disappointment with the ODAC outcome and will work with the FDA to complete the review of the sintilimab application.
Lilly also announced an agreement to supply the U.S. government up to 600,000 doses of its investigational COVID drug bebtelovimab for at least $720 million, if granted emergency approval by the FDA. Lilly has filed an application seeking emergency approval for bebtelovimab for mild-to-moderate COVID-19.
In January, the FDA revised the emergency use authorization (EUA) for Lilly’s already approved antibody cocktail medicine for COVID-19, bamlanivimab plus etesevimab, limiting their use in patients who have been infected or exposed to a coronavirus variant that is susceptible to these treatments.
The FDA’s decision to limit the authorized use of bamlanivimab/etesevimab was based on data, which showed that these treatment options are highly unlikely to be effective against the Omicron variant in the United States. However, Lilly said that an authentic virus analysis of bebtelovimab demonstrated that bebtelovimab neutralizes all known variants of concern, including Omicron.
The NYSE ARCA Pharmaceutical Index declined 0.9% in the last five trading sessions.
Here’s how the eight major stocks performed in the last five trading sessions.
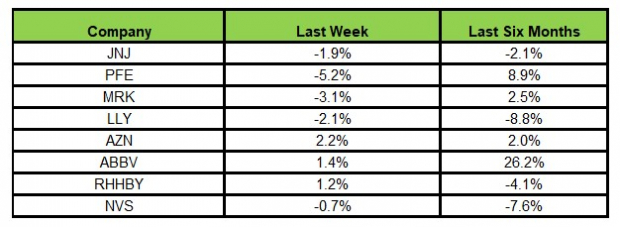
Image Source: Zacks Investment Research
In the last five trading sessions, AbbVie rose the most (2.2%) while Pfizer declined the most (5.2%).
In the past six months, AbbVie has recorded the maximum gain (26.2%) while Lilly declined the most (8.8%)
(See the last pharma stock roundup here:
MRK, LLY, ABBV Post Q4 Earnings, PFE Ends Vupanorsen Program
)
What’s Next in the Pharma World?
Watch out for regular pipeline and regulatory updates next week.
Just Released: Zacks Top 10 Stocks for 2022
In addition to the investment ideas discussed above, would you like to know about our 10 top picks for the entirety of 2022?
From inception in 2012 through 2021, the
Zacks Top 10 Stocks
portfolios gained an impressive +1,001.2% versus the S&P 500’s +348.7%. Now our Director of Research has combed through 4,000 companies covered by the Zacks Rank and has handpicked the best 10 tickers to buy and hold. Don’t miss your chance to get in…because the sooner you do, the more upside you stand to grab.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days.
Click to get this free report









